General Fish Health Management Info
NRAC Publication No. 111-2012

University of Maryland, 2113 Animal Science Building
College Park, Maryland 20742-2317
Telephone: 301-405-6085, FAX: 301-314-9412
E-mail: ssadams@umd.edu
Web: https://ansc.umd.edu/
General Fish Health Management
Paul R. Bowser, Professor of Aquatic Animal Medicine, Aquatic Animal Health Program Department of Microbiology and Immunology, College of Veterinary Medicine
Cornell University, Ithaca NY 14853-6401
AQUACULTURE is the production of fish and other aquatic organisms under controlled or semi-controlled conditions. For commercial success, an aquaculture operation must maintain fish at densities that greatly exceed those normally found in nature. Under these conditions,
fish must not only survive, but grow rapidly. Regardless of the culture system used (e.g., ponds, raceways, reuse systems, cages) it is imperative that the culturist maintain an environment conducive to good fish health.
A wide variety of pathogens (e.g. viruses, bacteria, parasites, fungi) can and do infect fish. Most diseasecausing agents are naturally present in low numbers and normally do not cause problems. The natural defense mechanisms of fish (i.e. undamaged skin, mucus covering the skin, and various components of the immune system) keep disease-causing agents in check. However, when fish already crowded in culture operations are further stressed (e.g., by low dissolved oxygen, nutritionally inadequate feeds, excessive handling) their natural disease defense systems may be weakened and their ability to protect against infectious diseases may be reduced. Disease induced catastrophic mortalities are frequently the result of, and response to, a stressful experience. Most disease problems can be avoided with proper management.
Avoidance of Disease
The aquatic environment provides a unique and effective means for the transmission of pathogens. Maintenance of suitable water quality greatly reduces the various stressors to which fish are exposed, which in turn reduces the likelihood of a disease problem. Critical water quality parameters include temperature (particularly sudden and dramatic shifts), dissolved oxygen, pH, alkalinity, hardness, nitrogenous wastes (un-ionized ammonia (NH3)); nitrite (NO2- anion), and potentially toxic substances (e.g., heavy metals, pesticides, carbon dioxide (CO2)). Many of these parameters are interrelated; for instance, as pH and temperature increase the proportion of total ammonia nitrogen (TAN) in the toxic un-ionized form (NH3) increases. Water quality should be monitored frequently and corrective measures initiated if conditions become stressful (e.g., dissolved oxygen below 3 ppm for warm water fishes or 5 ppm for cold water fishes, un-ionized ammonia in excess of 0.02 ppm is toxic to some fish species). The frequency of water quality monitoring will differ with different types of production systems and with the specific parameter being monitored. Dissolved oxygen in a pond can vary by a considerable amount during the day and is often measured several times during a 24-hour period. Hardness and alkalinity will typically be stable in a pond system, while it will change, but over a period of months in a closed recirculation system. The aquaculturist should discuss water quality monitoring with their aquaculture extension agent and develop a plan that is specific for their system
Use of a high quality feed provides fish with the nutrients that they need to remain healthy and to grow rapidly. Fish which are fed a nutritionally complete diet are better able to cope with stress and to resist disease. Culturists should remember that even high quality feeds will deteriorate if improperly stored or kept too long. Feed should be purchased from a reputable supplier, stored in a cool and dry place, and used in a timely manner (typically within 90 days or longer per the manufacturer’s instructions).
Light (excessive or rapid changes in intensity), noise and other disturbances can stress fish and should be minimized. Routine maintenance, stocking and harvesting require that fish be handled. When fish are removed and processed (e.g., weighed, sorted, transported) they compensate physiologically. To reduce the trauma of handling make sure all necessary materials (e.g., nets, hauling tanks, weighing scales) and adequate personnel are immediately available. Use of salt (1,000–2,000 ppm) in transport tank water can reduce stress when fish are moved (see the following section on Treatment of Diseased Fish for precautions). The transport water must also be properly aerated or oxygenated. Handle the fish gently and for as short a time as possible. If possible, do not handle fish that are already stressed or when environmental conditions are marginal (e.g., too hot, too cold, inhospitable).
Responding to Disease Problems
Regardless of how careful you are, if you culture fish long enough you will inevitably encounter a disease problem. When a disease problem develops, a quick and effective response is essential. There is no better preparation than to know your fish and your fish health specialist
Under routine aquaculture conditions, healthy fish display “normal” behavior. Fish feed vigorously when food is presented or shortly thereafter. In ponds, cages and raceways, fish are usually invisible, except when feeding. Therefore, it is important for the aquaculturist to note the feeding behavior of the fish being cultured even when automatic feeders are used. A reduced feeding activity should serve as notice to the aquaculturists that immediate further investigation of the cause is warranted. In raceways, fish normally swim leisurely, either enmasse or singly, depending on the species. Distribution in raceways varies for species, but is usually constant (e.g., some species prefer covered areas and others prefer uncovered areas, some concentrate toward the water inflow and others are more randomly distributed). As a culturist, you should become familiar with the normal behavior of your fish. If their behavior changes (e.g., they stop feeding, swim near the water surface, dart or scratch on objects), something has occurred and you need to find out what. The first response to a disease is abnormal behavior; to recognize what is abnormal, you must first be familiar with what is normal.
As previously mentioned, routine monitoring of water quality in a production system is imperative. When abnormal behavior is observed, culturists should check their water quality (e.g., dissolved oxygen, nitrogenous wastes, temperature). If water quality is a problem (e.g., low dissolved oxygen, high un-ionized ammonia) then corrective measures should be initiated. If abnormal behavior persists for several days or mortalities are observed, culturists should seek professional assistance. Water quality data should be provided to the diagnostic laboratory as well as information concerning the fish in question (e.g., symptoms, number of fish afflicted, when condition appeared). The following sections describe what you should do and who you should contact. Become familiar with the process, have the necessary supplies available, and know your diagnostic personnel and where they are located before a problem arises so you can address quickly and effectively the situation before a catastrophic die-off occurs.
Shipping Fish to a Disease Diagnostic Laboratory
Before shipping any fish, telephone your disease diagnostic laboratory (a list of facilities in the Northeastern United States is included at the end of this publication). Describe to the laboratory personnel the disease signs that you have observed and determine how they want you to ship your fish. Don’t ship fish without first consulting with laboratory personnel and do not expect a diagnosis over the telephone.
By informing the laboratory personnel of your problem and answering accurately their questions you can facilitate a rapid and accurate diagnosis. The more information that you can provide to the diagnostic laboratory, the better the evaluation of your disease case. Laboratory personnel will recognize that you are in a difficult situation and they will be anxious to help. Some general guidelines in specimen collection, preparation, and shipment follow.
I. The Specimen
The quality of specimen submitted to a fish disease diagnostic laboratory influences the ability of the fish health specialist to provide you with a diagnosis and recommendation for corrective action. Live fish showing the disease signs in question are preferred and should be collected with minimal stress. Dead fish are of little value for disease diagnosis; because:
a) Fish decompose very rapidly once death occurs. If the disease in question was caused by a pathogenic bacterium, other bacteria that take part in the normal decomposition process can quickly overgrow the pathogen and make its identification difficult or impossible.
b) Parasites require a live host for survival. When a fish dies, the parasites will often quickly leave the fish in search of another live fish upon which to live.
c) Viruses also require a live host in which to live. Once the fish dies the viruses will survive for only a limited period of time, sometimes only a few hours.
d) The time the fish has been dead is often impossible to know. Time is often wasted by providing a specimen that, although it may appear “fresh dead”, is actually unsuitable for processing. The extra effort to collect live fish that show the clinical signs of the disease in question is time well invested.
It is best to collect 3-5 living fish that show the signs of the disease and submit them to the fish health specialist to maximize the probability of identifying the causative agent.
II. Packing and Shipping the Specimens
The best possible way to transport sick fish to a disease diagnostic laboratory is for the culturist to bring them to the laboratory alive. This will provide the fish health specialist with the best specimens and the opportunity to obtain additional information from the culturist regarding the circumstances surrounding the mortalities. The culturist should also bring a water sample from the culture system if requested. This water sample should be collected in a clean container that can be capped tightly. If a chemical contaminant is suspected, the aquaculturist should bring this to the attention of the diagnostic laboratory. It is likely that you will be referred to a toxicology laboratory that specializes in chemical analyses. You will receive special instructions from the toxicology laboratory regarding how to collect samples for evaluation. If the culturist cannot transport the specimens and the fish must be shipped, the following methods (from most desirable to least desirable) can be used:
a) Live fish
b) Iced fish
c) Frozen fish
d) Formalin fixed fish
Most diagnostic laboratories prefer specimens that have not been preserved in formalin because the formalin will also fix (kill) pathogenic or disease causing microorganisms. Reaching a diagnosis may depend on the ability of the diagnostic laboratory to culture a bacterium or virus from the fish. This can be done only if the bacteria or viruses are not killed. In addition, a very important aspect of recommending a treatment for bacterial diseases is to determine the antibiotic resistance of the bacterium. The diagnostician must culture the microorganism in the laboratory to determine its resistance pattern. Formalin fixed materials can yield important diagnostic information following histological examination, but because of its limitations, most diagnostic laboratories prefer materials from which they can culture pathogenic microorganisms. The issue of formalin fixed specimens should be discussed with the diagnostic laboratory with which you will interact before specimens are treated and shipped.
Fish shipped by any of the above methods should be collected alive from those fish in the system that show signs of the disease. It is best to collect the fish with a net or trap. Capture of fish by rod and reel will select individual fish that are still feeding actively. One of the first behavioral signs of many diseases is that fish stop feeding. Thus, collecting fish with a rod and reel will select the healthiest fish in the population and accurate diagnosis of the disease may not be possible as these fish may not yet be infected.
A. Live Fish
- Obtain a strong, waterproof, insulated shipping container (e.g., a disposable styrofoam cooler in a sturdy cardboard box).
- Fill a heavy-duty plastic bag approximately 1/3 full of clean water from the culture facility. It is far preferable to use a “double bag system” (a bag within a bag) to guard against leakage during transport. Place the bag in the shipping container and add the fish. Fill the bag with pure oxygen or air. Seal the bag by twisting the open end tightly shut and securing it closed with several heavy-duty rubber bands or plastic tie-downs. An air tight seal is essential.
- Place 3-5 pounds of crushed ice or ice packs in a strong plastic bag, seal the bag as described above, and place it in the shipping container next to the bagged fish.
- In a separate, small plastic bag place a note preferably written in pencil and not ink that includes your name, address, telephone number, and information describing the fish and the culture system from which they came (e.g., why you suspect a disease; number of mortalities and their appearance; approximate size of diseased fish relative to other fish being cultured; when and how the shipped fish were collected; stock density; any known water quality parameters). Place the bagged note inside the shipping container.
- Seal the shipping container. Be sure to indicate which end is “up” and that live fish are enclosed.
CAUTIONS: Take extra care to make sure the container won’t leak. “Double bagging” is well worth the extra effort. Ship via a carrier that can provide overnight delivery. Many facilities will insist that an overnight shipment only be initiated on a Monday, Tuesday or Wednesday. An overnight shipment on a Thursday may not meet a Friday delivery schedule and could sit in transit because some diagnostic laboratories do not have weekend delivery service. This may result in the need for a second shipment, more expense and lost time. It is always best to contact your fish diagnostic laboratory prior to any shipment and to coordinate the receipt of the fish with them. A call afterwards to confirm fish arrival is prudent.
B. Iced Fish
- Obtain a strong, waterproof, insulated shipping container (e.g., a disposable styrofoam cooler in a sturdy cardboard box).
- Place each fish in a separate plastic bag and seal the bag. Live fish can be euthanized with an overdose of Finquel or Tricaine-S (Tricaine Methanesulfonate, MS222) that is buffered with an equal weight of sodium bicarbonate (dose: 250 mg/L each of MS222 and sodium bicarbonate).
- Place a larger, strong plastic bag (again, consider “double bagging”) in the shipping container and fill the bag with 2-4 inches of crushed ice.
- Place the individually bagged fish on the crushed ice in the larger bag and cover them with an additional 2-4 inches of crushed ice. Seal the larger bag by twisting the open end shut and securing it closed with several heavy-duty rubber bands or plastic tie-downs. An air tight seal is essential.
- In a separate, small bag place a note preferably written in pencil and not ink that includes your name, address, telephone number, and information describing the fish and the culture system from which they came (e.g., why you suspect a disease; number of mortalities and their appearance; approximate size of diseased fish relative to other fish being cultured; when and how the shipped fish were collected; stock density; any known water quality parameters). Place the bagged note inside the shipping container.
- Seal the shipping container. Be sure to indicate which end is “up” and that iced (perishable) fish are enclosed.
CAUTIONS: Adequate amounts of crushed ice, usually 10-15 pounds, will be satisfactory to keep the fish chilled during shipment. Ship via a carrier that can provide overnight delivery. Check with your Fish Disease Diagnostic Laboratory regarding the specific day on which they receive shipments. See CAUTIONS under “B. Live Fish” that are provided above.
C. Frozen Fish
- Obtain a strong, waterproof, insulated shipping container (e.g., a disposable styrofoam cooler in a sturdy cardboard box).
- Place each fish in an individual plastic bag and seal the bag. Freeze the fish in the individual plastic bags. Live fish can be euthanized with an overdose of Finquel or Tricaine-S (Tricaine Methanesulfonate, MS222) that is buffered with an equal weight of sodium bicarbonate (dose: 250 mg/L each of MS222 and sodium bicarbonate).
- Place a larger, strong plastic bag (consider “double bagging”) in the shipping container and fill the bag with 2-4 inches of crushed ice.
- Place the individually bagged, frozen fish on the crushed ice. Cover the fish with additional crushed ice and tightly seal the bag by twisting the open end shut and securing it closed with strong rubber bands or a plastic tie-down.
- In a separate, small bag place a note preferably written in pencil and not ink that includes your name, address, telephone number, and information describing the fish and the culture system from which they came (e.g., why you suspect a disease; number of mortalities and their appearance; approximate size of diseased fish relative to other fish being cultured; when and how the shipped fish were collected; stock density; any known water quality parameters). Place the bagged note inside the shipping container.
- Seal the shipping container. Be sure to indicate which end is “up” and that frozen (perishable) fish are enclosed.
CAUTIONS: Adequate amounts of crushed ice, usually 10-15 pounds, will be satisfactory to keep the fish chilled during shipment. Ship via a carrier that can provide overnight delivery. Check with your Fish Disease Diagnostic Laboratory regarding the specific day on which they receive shipments. See CAUTIONS under “B. Live Fish” that are provided above.
A. Formalin Fixed Fish
- Make a 10% formalin solution. Neutral buffered formalin is best. Under practical field conditions, water from the culture facility will usually provide adequate buffering capacity to the solution. (To prepare the desired formalin solution, mix 9 parts water with 1 part formalin).
- Live fish can be euthanized with an overdose of Finquel or Tricaine-S (Tricaine Methanesulfonate, MS222) that is buffered with an equal weight of sodium bicarbonate (dose: 250 mg/L each of MS222 and sodium bicarbonate).
- It is important that the fish be rapidly “fixed” by the formalin so that the quality of tissue preservation will yield useful information. Normally, formalin can rapidly fix tissues that are less than ½ inch thick. For this reason, the abdomen of larger fish must be opened for its entire length with one continuous cut. In addition, it may be necessary to remove the operculum from at least one side of the fish to ensure that the gills on that side are fixed. Most fish disease diagnostic laboratories will prefer to have the entire fish shipped rather than a limited number of tissues or organs. An “apparently normal organ” may yield valuable diagnostic information when examined microscopically. It is also important that adequate amounts of formalin be used to preserve the tissues. As a general rule, the ratio of formalin solution to tissue must be 10:1 by weight or volume (i.e. 1000 mL of the formalin solution: 100 gm fish). As mentioned above, it is important that you discuss the potential of shipping formalin fixed specimens with the fish diagnostic laboratory with which you interact.
- The container with the formalin, fish or tissue must be tightly sealed. Care should be taken to prevent breakage of the container. Glass containers can break and should be avoided. Use plastic bottles such as empty, clean food containers (e.g., peanut butter, mustard, salad dressing — use food service size for large fish) or containers obtained from scientific supply companies. As an alternative, the specimen could be fixed for 24-48 hours, the formalin poured off and the specimen(s) could be wrapped in gauze or paper towels that have been moistened with the buffered formalin. The specimens could then be packaged as above or placed in multiple strong ziplock plastic bags.
- The sealed container should be placed in a plastic bag with absorbent material and the bag should then be placed in a shipping container filled with styrofoam pellets or other suitable packing material. Care should be taken to prevent breakage.
- In a separate, small bag place a note preferably written in pencil and not ink that includes your name, address, telephone number, and information describing the fish and the culture system from which they came (e.g., why you suspect a disease; number of mortalities and their appearance; approximate size of diseased fish relative to other fish being cultured; when and how the shipped fish were collected; stock density; any known water quality parameters). Place the bagged note inside the shipping container.
- Seal the shipping container. Be sure to indicate which end is “up” and that the required US DOT safety labels are properly placed on the outside of the package.
CAUTIONS: Formalin is irritating and toxic. Provide for good skin and eye protection as well as use of a chemical hood or good room ventilation when using this chemical. A good practice when handling formalin or any other potentially irritating or toxic chemical is to use rubber gloves to protect the hands, goggles to protect the eyes, and a breathing mask to reduce inhalation of the chemical.
III. Summary
The manner in which your fish specimens are prepared and shipped will influence what information can be obtained when the specimens are examined by the fish health specialist. Working with a live fish will provide the diagnostician with the best opportunity to gain useful information regarding the fish disease. The fish can be examined for live parasites. Identification of living parasites can be aided by observing and characterizing their movement. Microorganisms (bacteria and viruses) can be cultured from specimens that are delivered to the diagnostic laboratory alive and the sensitivity of bacterial pathogens to potential treatment chemicals can be determined. A histopathological examination can be performed on properly prepared organs and tissues. Tissue changes that are indicative of disease can be identified. It is critical that the tissues be preserved properly to insure that they represent the disease process and not decomposition after death.
Specimens that are shipped on ice or frozen have some important diagnostic limitations. Living parasites may or may not be present on iced fish. Evaluation of iced tissues subsequently processed for microscopic 7 examination (histopthology) may not yield reliable information, depending upon the length of time the specimens were maintained in an iced condition. The limitations of frozen fish are greater. When frozen fish are thawed, the shearing action of melting ice crystals will destroy parasitic protozoa, making their identification very difficult if not impossible. The thawing of frozen tissues will also create a great deal of tissue damage, making tissues of limited or no use for histopathology. Iced and frozen specimens are normally satisfactory for the culture of bacteria and viruses.
Specimens shipped in formalin are useful for histopathological examination as long as the specimens have been carefully preserved prior to shipment. As mentioned above, the diagnostician must be able to detect tissue changes that are caused by the disease process and not the result of decomposition after death. This can only be done with a carefully processed sample. The formalin fixed sample may not yield the identity of the disease organism and it will not provide information regarding the antibiotic sensitivity of a bacterium.
The impact of fish handling and preservation on disease diagnosis can be summarized:

As stated earlier, the methods of shipment for fish disease diagnosis, from most desirable to least desirable are:
- Live fish
- Iced fish
- Frozen fish
- Formalin fixed fish
Treatment of Diseased Fish
Once a diagnosis has been made, the diagnostic laboratory will contact the culturist and identify the disease as well as recommend an appropriate and approved treatment or action. In certain cases a change in management is necessary. In other situations it is necessary to add an antibiotic to the feed (for internal bacterial infections) or a chemical to the water (usually for external parasite infestations). Regardless of diagnosis, it is extremely important that the aquaculturists follow closely the recommendations of the laboratory or the fish health professional and take appropriate precautions before any disease treatment is applied. For chemicals added to the water, it is usually a good precaution to test the treatment on a small group of fish before applying it to the entire unit.
Over the years, four cardinal rules of fish disease treatments have evolved:
a. Know your fish
b. Know your water
c. Know your chemical
d. Know your disease
The culturist must know his/her fish. What is their normal behavior, what conditions are likely to stress them, and to what diseases are they most susceptible. Some chemicals are safe and legal to use on certain species and ages of fish, but they may not be appropriate or approved for your fish.
In the United States, the U.S. Food and Drug Administration (FDA) has legal authority over the use of therapeutants or other compounds used for medical purposes in humans and animals. For fish, those compounds that are approved for use have a “label” which can be thought of as a statement that the compound may be used for specific purposes. The label will specify (1) the dose of the compound to be used, (2) the disease being treated, (3) the species of fish being treated and (4) a withdrawal time for the therapeutant if the fish is destined for human consumption. The aquaculturist must follow the specifics of a label for any therapeutant. There are certain instances when a therapeutant may be used for purposes beyond those listed on the label, but those uses must only be performed under the supervision of a licensed veterinarian.
No one chemical is appropriate for all diseases or situations. For instance, an antibiotic may be very effective in the treatment of certain bacterial infections, but is of little use if the disease is caused by a protozoan parasite. In addition, an antibiotic that is effective in treating one type of bacteria may be useless against other types of bacteria. All chemicals have precautions and considerations associated with their use. If an aquaculturists has no experience with a particular chemical, a small group of fish should be treated first, as a test before the entire lot is treated, to avoid potentially heavy losses due to toxicity associated with overtreatment. Extreme caution should be practiced when applying any chemical treatment. Water quality influences the toxicity of certain chemicals and is adversely affected by some chemicals. The culturist should be knowledgeable of the water quality in the culture facility. Of particular interest are dissolved oxygen, alkalinity, and the amount of organic material in the water.
Units
Units of measure used in this Publication are primarily metric. Concentrations of chemicals are commonly expressed in terms of milligrams (mg) per liter (L) or parts per million (ppm). When making a chemical application in freshwater these two terms are functionally equivalent. One liter of water weighs 1 kilogram = 1000 grams = 1,000,000 milligrams and 1 mg/ l,000,000 mg (or 1 L) = 1 ppm. The aquaculturist is also referred to the “Disease Treatment Calculator” prepared by the Fish Culture Section of the American Fisheries Society. This calculator can be found at:
Antibacterial compounds are added to the feed as a treatment for systemic (internal) bacterial infections. They are commonly applied as rates. A generic expression is the weight of antibacterial compound per weight of fish per day for a specified number of days. This may be in terms of mg drug/kg fish weight/day. Historically, some antibacterial treatment rates have been expressed as a combination of English and metric units (e.g., g (gram) drug/lb fish weight/day).
A list of conversion factors are provided at the rear of this publication to assist with calculations.
Aquaculture Chemicals
Below are brief descriptions of some commonly used aquaculture chemicals and precautions/ considerations associated with their use. It should again be emphasized that the aquaculturists must be aware of the legal status of using any chemical. A good practice is to maintain only those chemicals that have been “labeled” authorized by the FDA for use in treating disease in fish. This means that they have gone through extensive testing and have been found to be safe and effective for the purpose(s) and treatment regimens stated on the label. Essentially, they can be thought of as being approved by the FDA and are legal to use.
A second category of drugs are those considered by the FDA to be “Low Regulatory Priority.” These are drugs that have not gone through the extensive testing of labeled drugs and thus they are not Labeled for use in fish (i.e. they are not approved). However, there is sufficient scientific information and historical knowledge on the safe use of these compounds that the FDA considers them to be of a low priority for enforcement action. It is important to realize that these are not approved and the FDA may choose to take enforcement action on a case by case basis. But as long as these compounds are used in a manner consistent with the Low Regulatory Document such a regulatory action is not likely. An aquaculturist should only maintain compounds with specific approval for aquaculture uses at the production facility. The presence of non-approved chemicals at an aquaculture facility may imply their use to an inspector even if they are never used. Regulations concerning approved chemicals for use in aquaculture are continuously being updated. The most current information may be found on the Web site of the Center for Veterinary Medicine (CVM), FDA.
As of 2012, the Aquaculture Page of the CVM FDA web site contains links to many guidance documents of value to the aquaculturist:
http://www.fda.gov/AnimalVeterinary/DevelopmentApprovalProcess/Aquaculture/default.htm
The following page provides the most current list of all drugs approved for use in aquaculture:
http://www.fda.gov/AnimalVeterinary/DevelopmentApprovalProcess/Aquaculture/ucm132954.htm
The following page provides the most current list of Low Regulatory Priority drugs and the conditions under which they can be used:
http://www.fda.gov/downloads/AnimalVeterinary/GuidanceComplianceEnforcment/PoliciesProceduresManual/UCM046931.pdf
Additional information may be found in the FDA, CVM document titled: Fish and Fisheries Products Hazards and Controls Guide - Fourth Edition. Chapter 11 of that document, Aquaculture Drugs, contains an excellent summary of the currently available drugs for use in aquaculture. The Fish and Fisheries Products Hazards and Controls Guide - Fourth Edition may be found at:
Fish Disease Treatment Drugs that are FDA Approved
a) Terramycin (Oxytetracycline hydrochloride) is an antibiotic used to treat systemic (internal) bacterial infections. It is approved by the U. S. Food and Drug Administration (FDA) for the treatment of bacterial hemorrhagic septicemia caused by Aeromonas liquifaciens and pseudomonas disease in channel catfish and for the treatment of ulcer disease caused by Hemophilus piscium and furunculosis caused by Aeromonas salmonicida, bacterial hemorrhagic septicemia caused by Aeromonas liquifaciens, pseudomonas disease and for the control of coldwater disease associated with Flavobacterium psychrophilum in salmonids. It is used as a feed additive at a rate of 2.5 grams of drug (active ingredient)/100 pounds of fish weight/day for 10 days. A 21-day withdrawal period is required before the fish may be slaughtered and used for human consumption.
b) Romet-30 is a combination of two antibacterial drugs that has FDA approval for the treatment of furunculosis in salmonids and enteric septicemia in channel catfish. In both cases it is used as a feed additive at a rate of 50 milligrams drug (active ingredient)/kilogram of fish weight/day for 5 days. A 42-day withdrawal period is required for salmonids and a 3-day withdrawal period is required for channel catfish before the fish may be slaughtered and used for human consumption.
c) Aquaflor is the most recently labeled antibacterial drug available for use in fish. It is only available under a veterinary feed directive (VFD) issued by a licensed veterinarian in the course of the veterinarians’ professional practice. It is labeled for the following uses: in channel catfish for the treatment of Edwardsiella ictaluri infection and in salmonids for Aeromonas salmonicida infection and Flavobacterium psychrophilum infection through the use of a dose of 10 mg per kg of fish per day for 10 days; in catfish, tilapia, striped bass trout and all other freshwater-reared finfish at a dose of 10 – 15 mg/kg body weight/day for 10 days for the control of mortality due to columnaris disease associated with Flavobacterium columnare; in all freshwater-reared warmwater finfish at a dose of 15mg/kg for 10 days for the control of mortality due to streptococcal septicemia associated with Streptococcus iniae. The withdrawal time in for all fish is 15 days prior to slaughter.
d) Formalin-F, Paracide-F, Parasite-S® (formalin) is approved for use in the treatment of several external parasites. It is commonly used as an indefinite pond treatment at 15-25 "liters per liter ("L/L). In more limited quantities of water (ie. raceways, tanks, aquaria) formalin may pose a risk of low dissolved oxygen in the water. The formalin will remove 1 mg/L (ppm) dissolved oxygen for every 5 mg/L (ppm) of formalin used as a treatment. Therefore, aeration must be provided or a different treatment should be used. Formalin must be stored at temperatures above 40° F; below that temperature it will decompose and a white precipitate will be seen in the bottom of the container.
e) 35% Perox-aid (Hydrogen Peroxide) is approved to control mortality due to saprolegnia fungus in freshwater fish eggs at 500 – 1000 mg per liter (mg/L) of cultured water for 15 minutes in a continuous flow system once per day on consecutive days or alternate days until hatch for all coldwater and coolwater species of freshwater-reared finfish eggs or 750- 1000 mg/L for 15 minutes in a continuous flow system once per day on consecutive days or alternate days until hatch for all warmwater species of freshwater-reared finfish eggs.
35% Perox-aid is approved to control mortality in freshwater-reared salmonids due to bacterial gill disease associated with Flavobacterium branchiophilum. It is applied at 50-100 mg/L for 60 minutes once per day on alternate days for three treatments in continuous water supply or as a static bath.
35% Perox-aid is approved for the control in freshwater-reared coolwater finfish and channel catfish due to external columnaris disease associated with Flavobacterium columnare.
An initial bioassay on a small number of fish is highly recommended before treating an entire group of fish. Use with caution on walleye (Sander vitreus, previous name: Stizostedion vitreum).
SPECIAL NOTE:
a) Sulfamerazine is an antibiotic used at one time for the treatment of furunculosis in salmonid fishes. Old fish health literature implies that sulfamerazine is 9 an approved compound for use on food fish. The manufacturer has chosen to no longer market this product. The product was originally labeled under the trade name Sulfamerazine. It is inappropriate to substitute another sulfa drug for Sulfamerazine. The use of such substitute products is not legal.
Drugs that are considered Low Regulatory Priority (LRP) by the FDA:
a) Sodium Chloride (Salt; NaCl) is classified as a Low Regulatory Priority Drug for aquaculture use as an “osmoregulatory aid.” Salt can change the osmoregulatory balance (water balance) of freshwater aquatic organisms. It can control external parasitic protozoans by placing them in a condition of severe osmoregulatory shock. Care must be exercised to avoid overtreatment which will place the fish in the same condition of osmoregulatory shock. Sodium chloride is used as a 1,000 to 2,000 parts per thousand (ppt) concentration in water as an indefinite (long term) treatment or as a 3% concentration in water for 10-30 minutes (stop the treatment earlier if the fish show signs of stress).
Products found not to be Low Regulatory Priority but regulatory action deferred pending further study:
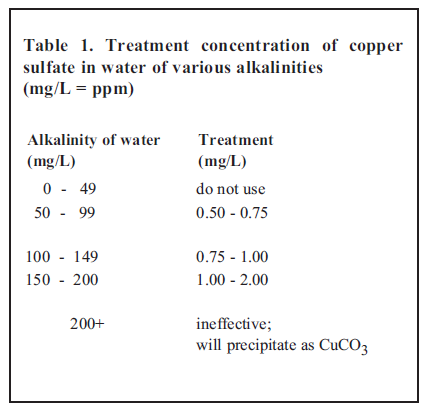
a) Copper Sulfate (CuSO4) has been used to treat a variety of external parasites of fish. It is also an effective algaecide (algicide), and can kill fish if used improperly. The relationship between toxicity of copper sulfate and alkalinity is very important (alkalinity is the total concentration of alkaline substances in the water expressed as equivalent calcium carbonate). In water with an alkalinity less than 50 milligrams per liter (mg/L), copper sulfate can be very toxic to fish and should not be used unless a bioassay has been run in the water first with a limited number of the fish to be treated. The general guidelines provided in Table 1 have been established for the use of copper sulfate.
Since copper sulfate is an algaecide (algicide), consideration must be given to dissolved oxygen in a pond to be treated. If a pond already has low dissolved oxygen, an alternate treatment should be used. Copper sulfate will only aggravate low dissolved oxygen problems by killing the primary source of oxygen (the algae) and by adding a large biological oxygen demand in the form of dead and decomposing algae.
b) Potassium Permanganate (KMnO4) has been used in aquaculture as an oxidizer and detoxified. It has been used effectively against a number of external disease organisms of fish, including the columnaris bacterium (Flavobacterium columnare) and the fungal pathogen Saprolegnia sp. The normal treatment is 2-8 milligrams per liter (mg/L), depending upon the amount of organic matter in the pond to be treated. Ideally, one would like to maintain a red color in the water for a 12-hour period to ensure an effective treatment. A preliminary test can be performed with a small volume of culture water to determine the appropriate dose for the system.
Calculation of Disease Treatments
The aquaculturist is again referred to the “Disease Treatment Calculator” prepared by the Fish Culture Section of the American Fisheries Society. This calculator can be found at:
Water treatments are based on water volume. A specified amount of chemical is added to a known quantity of water for a specified time. If too little chemical is added the treatment will be ineffective; if too much is added or if the fish are left in contact with the chemical too long, they may become stressed or die. Use care or sufficient mixing when adding chemicals or stock solutions to rearing units to avoid creating “hot spots” where fish encounter toxic concentrations.
Feed treatments and fish injections are based on fish weight. A specified amount of chemical is added to the feed or injected into the fish. Improper doses may result in an ineffective treatment or mortalities.
Aquaculturists should compute the volume of each culture unit (e.g., pond, tank, raceway) before a problem occurs, preferably when the system is designed or filled with water for the first time. The information should be stored so it is immediately available when needed. Practice calculations should be done so the culturist is comfortable and familiar with the computation procedure.
Comments on Scientific Names
Over the course of time new scientific information is always becoming available on various pathogens of fish. One area that is constantly changing, and can lead to confusion is the scientific names of fish and fish pathogens. In the above discussion of diseases and disease treatments, several of the names of fish pathogens have changed since the time the official documents were published. Since those documents were official legal documents, the names must stand as they were at the time of publication. Those organisms noted above that have undergone name changes are
Hemophilus piscium is now considered to be a variety of Aeromonas salmonicida, the bacterium causing furunculosis disease.
Aeromonas liquifaciens is now known as Aeromonas hydrophila and the disease is commonly called motile aeromonad septicemia.
Flavobacterium columnare (as listed above) is the current name for the bacterium causing columnaris disease. Older literature may refer to it as Flavobacterium columnaris, Flexibacter columnaris, Cytophaga columnaris, or Chondrococcus columnaris.
Sample Calculations
To provide the culturist with an opportunity to become familiar with the methodology used to calculate fish disease treatments, three hypothetical situations are presented on the following pages. For each example, there are several ways to correctly compute the amount of chemical to add or the drug to use. Calculations and steps are shown in detail for one method. 11 12
Example 1
You have a raceway with rainbow trout that are infected with the parasitic protozoan, Ichthyophthirius. You elect to treat with copper sulfate (CuSO4). The raceway contains 5,000 gallons of water with an alkalinity of 75 milligrams per liter (mg/L or ppm). How much CuSO4 would you use?
Computation steps:
- Examine Table 1 (found on page 10) and determine what concentration of CuSO4 should be added to the system to provide an appropriate and safe treatment. You know that the alkalinity is 75 mg/L. Therefore, an appropriate treatment concentration for CuSO4 is 0.5 mg/L (the lower concentration found in the above Table 1 was selected for this example).
- Determine the quantity of CuSO4 to be added to the raceway to achieve the 0.5 mg/L concentration.
-
Convert the volume of the raceway from gallons (gal) to liters (L). (5,000 gal) x (3.8 L per gal) = 19,000 L of water in the raceway
- Determine a correction factor (see insert box below) for the proportion of the chemical (CuSO4) that is the active ingredient. (Note: Copper Sulfate is typically sold as a pure chemical and contains no inert fillers. It is 100% active). (100%) / (100% active ingredient) = correction factor = 1.0
- Compute the amount of chemical (CuSO4) that should be added to the raceway, (volume of race way) x (dosage of CuSO4) x (correction factor) (19,000 L) X (0.5 mg/L CuSO4) X (1.0) = 9,500 mg CuSO4
- Convert milligrams (mg) CuSO4 to grams (g). (9,000 mg) / (1,000 mg/ 1.0 g) = 9.5 g CUSO4 added to the 5,000 gallon (or 19,000 L) raceway
NOTE: Because Ichthyophthirius has a complicated life cycle that must be considered in its treatment, applications of chemical are made every third day for 3 to 4 treatments. *Correction Factor: Percent active ingredient is the purity of the chemical or the amount that is effective in treatment of the disease. Copper sulfate is usually available in a 100% active form (pure copper sulfate), but many chemicals are not. If the chemical is not 100% active, a correction factor must be generated: 100% / percent active ingredient = correction factor The percent active ingredient information can be obtained from the label on the container in which the chemical was supplied or from personnel at the diagnostic laboratory.
Example 2
If alkalinity in Example 1 was below 50 mg/L, CuSO4 would not have been the treatment of choice due to potential toxicity to the fish (see Table 1 above). An alternative treatment would have been formalin.
Computation steps:
- Obtain from your diagnostic laboratory a recommended treatment concentration for formalin. An appropriate dose is 25 mg/L (or 25 ppm formalin)
-
Determine the quantity of formalin to be added to the raceway to achieve the 25 mglL (25 ppm) concentration.
- Convert the volume of raceway from gallons (gal) to liters (L). (5,000 gal) X (3.8 L per gal) = 19,000 L in the raceway
- Determine a correction factor for the proportion of chemical (formalin) that is active ingredient. NOTE: Although formalin is 37% formaldehyde gas dissolved in water, for fish treatment purposes formalin is considered to be 100% active. (100%) / (100% active ingredient) = correction factor = 1.0
- Compute the amount of chemical (formalin) that should be added to the raceway. (volume of raceway) X (dosage of formalin) X (correction factor) (19,000 L) X (25 mg/L formalin) X (1.0) = 475,000 mg formalin
-
Since formalin is a liquid it is desirable to convert milligrams (mg) to milliliters (mL). (475,000 mg formalin) / (1,000 mg / 1.0 g) = 475 g formalin 1.0 g formalin is approximately equal to 1.0 mL formalin, therefore 475 g formalin is approximately equal to 475 mL formalin to be added to the 5,000 gallon raceway
Example 3
You have catfish in a pond with a diagnosed bacterial (Aeromonas hydrophila) infection. The pond contains 100 fish that weigh an average of 2 pounds each, for a total of 200 pounds of fish. The bacterium is sensitive to Terramycin. How would you prepare the treatment?
Computation steps:
- Terramycin is frequently used at 2.5 grams (g) active ingredient per 100 pounds (lb) of fish per day for 10 days. The drug is mixed with the feed and fed to the fish.
-
Determine the quantity of Terramycin needed for the 10 day treatment.
- Determine a correction factor for the proportion of chemical (Terramycin) that is active ingredient*. NOTE: Terramycin premix is often supplied as a 50% active mixture. Always check the label of a specific package. (100%) / (50% active ingredient) = correction factor= 2.0
- Compute how much Terramycin is to be fed each day. (dosage of Terramycin) X (lb of fish in pond) X (correction factor) (2.5 g Terramycin / 100 lb fish) X (200 lb fish) X (2.0) = 10.0 g Terramycin per day
- Compute the quantity of Terramycin fed for 10 days. (10.0 g Terramycin / day) X (10 days) = 100.0 g Terramycin
-
Determine the amount of food that you will feed to the fish during the 10 day treatment period.
- The catfish that are infected might reasonably be fed at a rate of 1% of their body weight per day. (200 lb fish) X (1.0%/ 100%) = 2.0 lb food per day
- Compute feeding rate for 10 days. (2.0 lb food/ day) X (10 days) = 20.0 lb food
-
Prepare medicated feed and present it to the fish
Commercially prepared, medicated feeds are available and can be used. Alternatively, medicated feed can be prepared by mixing the 100 grams of Terramycin with 20 pounds of feed. The antibiotic may be mixed in a vegetable or fish oil. It is then spread on the feed pellets which become coated with a thin film of the antibioticlaced oil. Regardless of the source of the medicated feed, 2.0 pounds of that medicated feed are provided to the fish each day for 10 days.
The importance of obtaining an accurate diagnosis for a disease problem cannot be overemphasized. The success of any treatment is closely tied to knowing the condition being treated and being aware of any potential complications. Accurate diagnosis of a fish disease requires specialized technical skills and appropriate laboratory facilities. Fish disease diagnostic services are available from several sources in the northeastern United States. The aquaculturist should identify a laboratory 15 capable of providing fish disease diagnostic services as part of their initial organization and set-up. In some cases this may require contacting a facility in a neighboring state. The producer should make contact with that laboratory before a problem occurs to determine any specific instructions associated with the submission of fish for diagnostic evaluation. For instance, some laboratories can, due to their funding, only accept submissions from restricted geographical locations and some fish health research laboratories only accept diagnostic cases that are referred by other diagnostic laboratories.
Fish Disease Diagnostic Services in the Northeastern United States
Connecticut
Connecticut Veterinary Diagnostic Laboratory (CVMDL)
University of Connecticut
61 North Eagleville Rd., U-3089
Storrs, CT 06269
Contact: Sal Frasca
(860) 486-3738
CVMDL@uconn.edu
Fish Health Laboratory
Connecticut DEEP
Contact: Richard Van Nostrand
(860) 673-3695
richard.vannostrand@ct.gov
Delaware
(none known at this time)
Maine
Aqua Vet Group
350 Commercial Street
Portland, ME 04101-5597
Contact: Jason Collins
jason.collins@fishvetgroup.com
(207) 699-5902
Kennebec River Biosciences
41 Main St.
Richmond, ME 04357
www.kennebecriverbiosciences.com
Contact: Cem Giray
(207) 737-2637 (tel.)/(207) 737-4504 (fax)
Univ. of Maine Animal Diagnostic Laboratory
5735 Hitchner Hall
Orono, Maine 04469-5735
Contact: Deborah A. Bouchard
(207) 581-2767
deborah.bouchard@maine.edu
(Primarily for fish health research)
Maryland
(none known at this time)
Massachusetts
(none known at this time)
New Hampshire
New Hampshire Veterinary Diagnostic Laboratory
University of New Hampshire
129 Main St
Durham, NH 03824
Contact: Inga F. Sidor
inga.sidor@unh.edu
(603) 862-2726 main/(603) 862-2743 office (603) 862-0179 fax
New Jersey
NJ Public Health, Environmental and Agriculture Laboratory
3 Schwartzkopf Dr.
Ewing, NJ 08628
mailing address: P.O. Box 330 Trenton NJ 08625
Contact: Amar Patil (609) 671-6400 or (609) 406-6999
amar.patil@ag.state.nj.us
New York
Fish Disease Diagnostic Laboratory
Department of Microbiology and Immunology
College of Veterinary Medicine
Cornell University
Ithaca, NY 14853
Contact: Paul R. Bowser
prb4@cornell.edu
(607) 253-4029/(607) 253-4028 (Lab)
Pennsylvania
Animal Diagnostic Laboratory
Penn State University
University Park, PA 16802
(814) 863-0837
adlhelp@psu.edu
Rhode Island
Aquatic Diagnostic Laboratory
Department of Biology and Marine Biology
Roger Williams University
One Old Ferry Road, Bristol, RI 02809
Contact: Roxanna Smolowitz
401-254-3299
rsmolowitz@rwu.edu
Vermont
(none known at this time)
Washington, D.C.
(none known at this time)
West Virginia
(none known at this time)
Comprehensive directory of aquatic veterinarians and disease diagnostic laboratories: http://www.aquavetmed.info/
Acknowledgments
This publication is a revision of NRAC Bulletin 111- 1993 General Fish Health Management. The earlier version  benefitted from insightful criticisms from Drs. John A. Plumb and Wilmer. A. Rogers, Auburn University; Dr. Thomas E. Schwedler, Clemson University; Mr. Jerry Williamson, Farm Fresh Catfish Company; Dr. David Smith, Freshwater Farms of Ohio, Inc.; Mr. Gef Flimlin, New Jersey Sea Grant Marine Advisory Service; Mr. Donald Webster, University of Maryland - Cooperative Extension Service; and Dr. Michael Rice, University of Rhode Island. We express our appreciation for Drs. Meg Oeller, Jennifer Matysczak and Stacey Gore of the Center for Veterinary Medicine, U.S. Food and Drug Administration for helpful comments on our discussion of therapeutants in this current document. Additionally, extension representatives and/or specialists from each fish disease diagnostic facility listed in this publication provided input regarding the current availability of fish disease diagnostic services in their state.
benefitted from insightful criticisms from Drs. John A. Plumb and Wilmer. A. Rogers, Auburn University; Dr. Thomas E. Schwedler, Clemson University; Mr. Jerry Williamson, Farm Fresh Catfish Company; Dr. David Smith, Freshwater Farms of Ohio, Inc.; Mr. Gef Flimlin, New Jersey Sea Grant Marine Advisory Service; Mr. Donald Webster, University of Maryland - Cooperative Extension Service; and Dr. Michael Rice, University of Rhode Island. We express our appreciation for Drs. Meg Oeller, Jennifer Matysczak and Stacey Gore of the Center for Veterinary Medicine, U.S. Food and Drug Administration for helpful comments on our discussion of therapeutants in this current document. Additionally, extension representatives and/or specialists from each fish disease diagnostic facility listed in this publication provided input regarding the current availability of fish disease diagnostic services in their state.
This publication was supported by the Northeastern Regional Aquaculture Center through grant number 2007-38500-18589 from the Cooperative State Research Service, U.S. Department of Agriculture. Any opinions, findings, conclusions or recommendations expressed in this publication are those of the authors and do not necessarily reflect the view of the U.S. Department of Agriculture.
Some Useful Conversion Factors
1 ppm (mg/L) = 0.38 grams per 100 gallons of water = 3.8 milligrams per gallon of water = 0.0283 grams per cubic foot of water = 0.38 milliliters per 100 gallons of water = 2.72 pounds per acre-foot of water = 1 milligrams per liter of water = 1 grams per cubic meter of water = 0.001 milliliters per liter of water English : Metric Conversions 1 acre-foot = 43,560 cubic feet 1 acre-foot = 325,850 gallons 1 acre-foot of water = 2,718,144 pounds 1 cubic foot of water = 7.48 gallons 1 cubic foot of water = 62.4 pounds 1 cubic foot of water = 28.3 liters 1 cubic foot of water = 28.3 kilograms 1 cubic meter of water = 1,000 liters 1 cubic meter of water = 35.3 cubic feet of water 1 cubic meter of water = 2.203 pounds of water 1 gallon of water = 8.34 pounds 1 gram = 0.0352 ounces 1 kilogram = 2.2 pounds 1 pound = 454 gram 1 gallon = 3.785 liters 1 gallon of water = 3,785 grams 1 liter = 0.26 gallons 1 liter = 1,000 cubic centimeter 1 liter = 1,000 milliliters 1 liter of water = 1,000 grams 1 ounce (weight) = 28.4 grams 1 gallon = 128 fluid ounces 1 fluid ounce = 29.6 grams 1 inch = 2.54 centimeters 1 foot = 30.48 centimeters 1 cubic centimeter of water = 1.0 gram 1 cubic centimeter of water = 1.0 milliliter 1 hectare = 10,000 square meters 1 hectare = 2.47 acres 1 acre = 0.405 hectare 1 acre = 43,560 square feet Percent Solution For 1 percent solution add: 38 grams per gallon 1.3 ounces per gallon 38 cc per gallon 10 grams per liter 10 cc per liter Temperature Conversion Centigrade to Fahrenheit = (C X 9/5) + 32 Fahrenheit to Centigrade = (F - 32) X 5/9


