Optogenetic toolkit will shed light on cardiac, lung, blood, and immune disease
 New technologies involving optogenetic proteins, which use light to control and observe cells with unprecedented precision, have begun to illuminate processes underlying cellular behavior and the effects of cell and gene–based therapies. Cornell researchers are developing advanced forms of these proteins to form a toolkit to make them more widely available to scientists.
New technologies involving optogenetic proteins, which use light to control and observe cells with unprecedented precision, have begun to illuminate processes underlying cellular behavior and the effects of cell and gene–based therapies. Cornell researchers are developing advanced forms of these proteins to form a toolkit to make them more widely available to scientists.
With a five year $3.1 million grant from the National Heart, Lung, and Blood Institute, the team will develop the Cornell Heart, Lung, and Blood Resource for Optogenetic Mice (CHROMus), which will incorporate optogenetic proteins in mice and human stem cells. Scientists use such tools to control and observe how different types of cells function and interact.
“We will target these tools so that they can be combined to study diseases of the heart, lungs, vasculature, and blood,” said Dr. Michael Kotlikoff, the Austin O. Hooey Dean of Veterinary Medicine and at Cornell’s College of Veterinary Medicine and the project’s lead investigator. “Researchers will be able to use them to address a broad set of health issues, including heart attack, stroke, asthma, and immune diseases.”
Marrying optics and genetics, optogenetics enables scientists to use light to trigger and monitor the behavior of cells engineered to contain one or both of two types of designer proteins: effectors, which respond to light by activating the cell they are on, or sensors, which fluoresce when a cell has been activated.
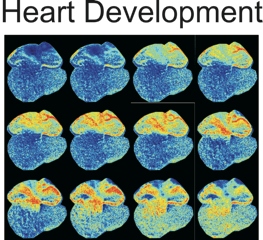 Effectors and sensors can be engineered into specific kinds of cells and color-coded, letting scientists non-invasively trigger one type to see how another type responds. One can see different cell types light up in living animals, giving direct insight into specific cells’ roles in complex biological systems.
Effectors and sensors can be engineered into specific kinds of cells and color-coded, letting scientists non-invasively trigger one type to see how another type responds. One can see different cell types light up in living animals, giving direct insight into specific cells’ roles in complex biological systems.
The lines of CHROMus mice developed in this project are designed to be easily crossed, creating a combinatorial platform that will allow scientists to customize sets of effectors and sensors, including new sensors from the Kotlikoff lab, into the specific cell types they want to study.
“For example, our lab is particularly interested in using these tools to study the control of blood flow to tissues—what happens before, during, and after major events like stroke and cardiac infarction, and how abnormal rhythms develop after heart injury,” said Kotlikoff. “Arrhythmias following a heart attacks are the single most common cause of acute death in the western world, and how they can be prevented requires a better understanding of how, why, and where they arise. Optogenetic tools let us look directly at relevant cells throughout the heart to determine their role in these dangerous and often fatal events.”
The tools will be designed to allow scientists to ask and answer similar questions related to vascular and lung diseases, such as the role of the immune system in asthma and stroke, and how therapeutic stem cells integrate within the tissue that they are designed to repair.
“This resource will allow investigators across institutions to use optogenetic tools in their work,” said Kotlikoff. “It will lower the barriers to using these technologies. The expertise to make these tools and use them in specific cells doesn’t often overlap— Cornell is well positioned to do this because we have an unusual combination of strengths and resources in genetics, optics, protein engineering, and molecular biology.”
The CHROMus website will freely distribute information on the tools under development and their availability for research: CHROMus.vet.cornell.edu.





