How a key cellular process in dog cancers might open new therapies for human breast cancer
A newly published study sheds light on how certain canine cancers suppress the immune system, and how those findings may advance treatments for human breast cancer. The research, published online on January 22 in Communications Biology has broad cross-species implications. “These findings reinforce that studying human diseases, including cancer, in companion animals can advance our understanding of the disease in both species,” said Kimaya Bakhle, a D.V.M.-Ph.D. program pathway student in Dr. Anushka Dongre’s lab and first author on the paper. “For example, by researching naturally-occurring canine mammary tumors, we were able to identify conserved mechanisms by which tumors evade the immune system, as well as a novel marker of malignancy.”
Dongre, assistant professor in the Department of Biomedical and Translational Sciences and corresponding author on the study, reiterated the benefit of canines as models for cancer research. “Dogs offer an incredibly valuable window into how cancers behave in real time."
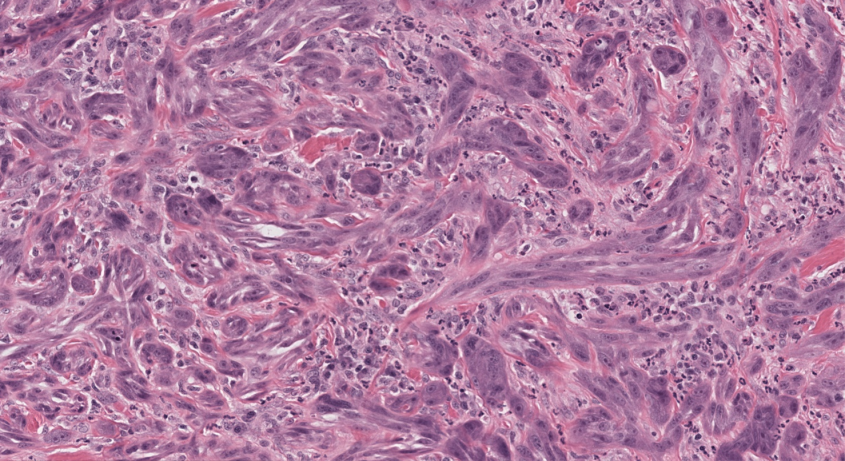
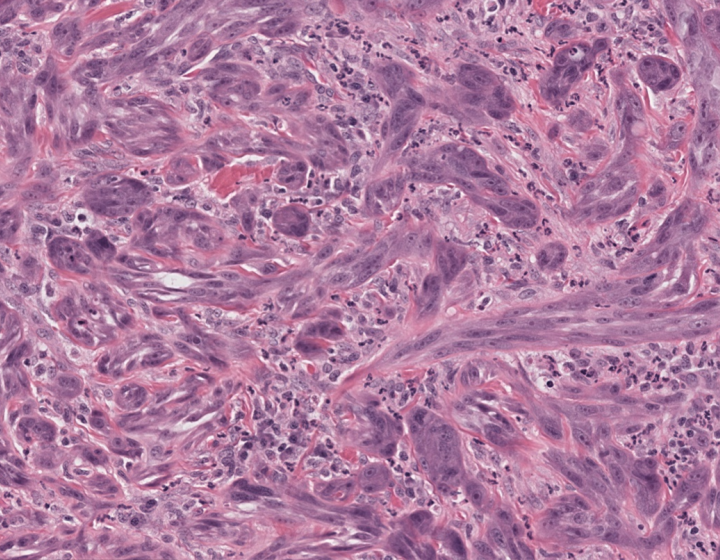
The work focuses on naturally occurring canine mammary carcinoma, which closely mirrors human breast cancer, and how it interacts with a biological program called epithelial–mesenchymal plasticity (EMP). It occurs when epithelial cells (found in the lining of various internal body surfaces) transform into mesenchymal cells, which can detach and travel around the body. Cancer can hijack this process for its own gain, promoting invasiveness, metastasis, chemotherapy resistance and immunity suppression, undermining the body’s ability to fight the cancer. “What we’re seeing is that when canine tumors activate EMP, they don’t just become more aggressive — they also actively shut down the immune response,” said Dongre.
A conserved mechanism across dogs, mice and humans
Previously, Dongre had demonstrated EMP‑driven immune suppression in a mouse model, but it remained unclear whether these findings extended to real-world canine tumors. The new study, led by Bakhle, provides that missing link, confirming that EMP activation in dogs attracts regulatory T‑cells and M2-like macrophages, which both dampen anti‑tumor immunity. “Seeing this same biology in mice, dogs and humans tells us we’re looking at a deeply conserved mechanism,” Dongre said. “If we can target EMP-driven immune suppression in dogs, those therapies could very well translate to people.”
CD109: A new player in immune suppression
The research team also identified CD109, a cell-surface glycoprotein, which plays a key role in EMP‑mediated immune suppression across dog, mouse, and human cancer models. While CD109 has previously been associated with promoting tumor growth, this is the first evidence connecting it to immune evasion in any species. “CD109 is known for its role in tumor formation,” Dongre said. “But its involvement in immune suppression was unexpected. This opens a completely new therapeutic avenue we hadn’t considered before.”
Immune suppression found across multiple canine cancers
The researchers also found that EMP‑associated immune suppression is not limited to mammary tumors. Dongre’s team also found that oral squamous cell carcinoma, urothelial carcinoma and pulmonary carcinoma had consistently high levels of immune-suppressing molecules across multiple tumor types. “The fact that we’re seeing the same immunosuppressive signals in several completely different cancers tells us the EMP program is playing a broad and powerful role,” Dongre said. “This doesn’t appear to be an isolated phenomenon.”
Implications for human breast cancer treatment
Comparative oncology researchers have long recognized dogs as a valuable model for human disease, given that they develop cancer naturally and share environmental exposures with people. Because canine mammary carcinomas closely mimic human breast cancer, these findings provide a potential framework for understanding why some tumors resist immunotherapy. “Human breast cancers with strong EMP signatures tend to be the ones that don’t respond well to immunotherapy,” Dongre said. “Our data suggest that part of the reason may be this EMP‑driven recruitment of suppressive immune cells. If that’s true, we may eventually be able to target these pathways to improve patient outcomes.”