2021-2022 Funded Research Projects
Is Campylobacter a hidden source of canine GI troubles?
 Dogs can have an upset stomach and diarrhea for many reasons—getting into the trash, a new type of kibble or too many table scraps. But Dr. Kevin Cummings suspects there may be another cause that veterinarians could be missing: a bacterium called Campylobacter jejuni, which is a common cause of food poisoning in people. With support from the Cornell Richard P. Riney Canine Health Center, Cummings is investigating whether the bacterium is an important cause of diarrhea in dogs and what factors might put a dog at risk of infection.
Dogs can have an upset stomach and diarrhea for many reasons—getting into the trash, a new type of kibble or too many table scraps. But Dr. Kevin Cummings suspects there may be another cause that veterinarians could be missing: a bacterium called Campylobacter jejuni, which is a common cause of food poisoning in people. With support from the Cornell Richard P. Riney Canine Health Center, Cummings is investigating whether the bacterium is an important cause of diarrhea in dogs and what factors might put a dog at risk of infection.
"Even from my days as a practitioner, I had this suspicion that we could be missing cases of C. jejuni in dogs," said Cummings, who is an associate professor of epidemiology at the Cornell College of Veterinary Medicine. "If that's the case, we're missing an opportunity to make an accurate diagnosis and optimize the therapeutic plan for the canine patient. Also, we may be unknowingly sending patients home with an infection that can be transmitted to people."
Veterinarians rarely test for C. jejuni, likely due to the added cost and the myriad other potential causes of diarrhea. The infection typically clears up on its own, but there is a risk the infected dog will pass the bacterium to humans or other animals. In recent years the Centers for Disease Control and Prevention (CDC) linked several cases of multidrug-resistant C. jejuni in people to infected pet store puppies.
To find out whether C. jejuni may be an under diagnosed cause of diarrhea in dogs, Cummings will be working with collaborators at the Cornell University Hospital for Animals to test stool samples from dogs with diarrhea and healthy dogs coming in for routine checkups. Their tests will show what proportion of dogs in each group are shedding the bacterium. They suspect that dogs with acute diarrhea will be more likely to harbor C. jejuni than healthy dogs. They also plan to examine any C. jejuni that they culture from stool samples to see if they are resistant to antibiotics.
Cummings will also try to identify factors that may increase a dog's risk of becoming infected, such as different types of diets, medications or visits to dog parks and doggy daycare facilities. "One big dietary factor that is on our mind is raw meat diets, which present a clear risk, not only for Campylobacter, but also Salmonella," he said.
Cummings hopes that his CHC-funded study will reveal potential risk factors so that veterinarians will have a better idea when to test a dog for C. jejuni. Additionally, owners will be able to limit their dog's exposure to prevent their pets—and themselves—from becoming infected.
Written by Patricia Waldron
Laying the groundwork to find new therapies for oral tumors
 Dogs with a type of mouth cancer called oral squamous cell carcinoma (OSCC) often undergo a disfiguring surgery that sometimes removes up to a quarter of the jawbone. Drs. Santiago Peralta and William Katt think there might be a better way. They are developing new models of OSCC with funding from the Cornell Richard P. Riney Canine Health Center to test out novel therapies in the lab that may one day be used in patients.
Dogs with a type of mouth cancer called oral squamous cell carcinoma (OSCC) often undergo a disfiguring surgery that sometimes removes up to a quarter of the jawbone. Drs. Santiago Peralta and William Katt think there might be a better way. They are developing new models of OSCC with funding from the Cornell Richard P. Riney Canine Health Center to test out novel therapies in the lab that may one day be used in patients.
"We deal with this type of tumor routinely in the clinic," said Peralta, an associate professor of dentistry and oral surgery at the Cornell University College of Veterinary Medicine. "Treatment options have been unchanged for a very long time and the surgical solutions are often devastating to the patients. With the advent of novel technologies and amazing expertise from Cornell, we realized we could join forces and come up with better ways to treat OSCC patients."
Katt, a senior research associate in Molecular Medicine at Cornell specializing in cancer drug discovery, met Peralta at a cross-campus conference. They decided to team up and apply their very different areas of expertise to treating oral tumors.
Even for humans with mouth cancer, there aren't many "gentle therapies," said Katt. "Canine oral cancer is thought to really well represent human oral cancer. So, we hope that anything we discover that works in canines could then be adapted to help human patients as well."
Scientists must screen potential new drugs in the lab for safety and effectiveness before giving them to patients. But for canine oral tumors, there are no screening tools available. To correct this problem, Peralta is taking cancerous tissues from his patients and growing them inside mice, while Katt is turning them into cell lines on plates in the lab. They aim to develop up to 10 mouse models with matching cell lines to represent the genetic diversity found in OSCC tumors.
Next, the team will test potential drugs in the cell lines and then in mice implanted with the tumors to identify ones that successfully shrink them. Ultimately, they hope to discover effective pharmacological approaches that can be tested further in clinical trials to treat dogs with OSCC. The work may one day result in fewer disfiguring surgeries.
Additional collaborators on this project include Rishi Puri, project manager of the PATh PDX Facility, which is developing the mouse tumor models; Jen Grenier, director of the Genomics Innovation Hub, who is helping with tumor sequencing efforts; and Gerald Duhamel, professor of anatomic pathology in Biomedical Sciences, who confirms that the tumors are in fact OSCC. Susan Garrison and Denise F. LaLonde-Paul from the Cornell Veterinary Biobank are helping to facilitate the collection and storage of samples.
"We are making excellent use of the Cornell facilities and the experts they have on hand to make sure everything is being done correctly," said Peralta. "This is critical for discovery."
Written by Patricia Waldron
Getting closer to the cause of Legg-Calve-Perthes disease
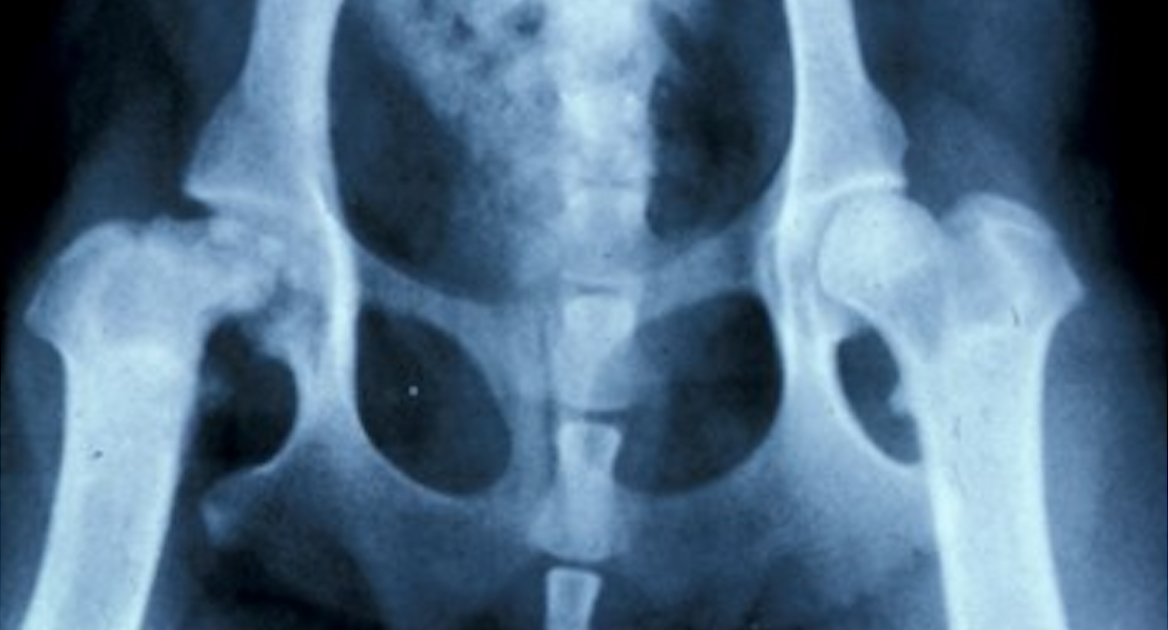 Legg-Calve-Perthes disease is a rare but devastating condition causing hip pain and lameness. Its cause is currently unknown, so Dr. Rory Todhunter is searching the canine genome for mutations that might be involved. The Cornell Richard P. Riney Canine Health Center is funding this work, which may one day lead to a genetic test that will prevent future dogs from being born with the disease.
Legg-Calve-Perthes disease is a rare but devastating condition causing hip pain and lameness. Its cause is currently unknown, so Dr. Rory Todhunter is searching the canine genome for mutations that might be involved. The Cornell Richard P. Riney Canine Health Center is funding this work, which may one day lead to a genetic test that will prevent future dogs from being born with the disease.
Legg-Calve-Perthes disease typically shows up in small dog breeds, striking when dogs are just months old. It occurs when the ball at the top of the femur loses its blood supply and breaks down inside the hip joint. The only solutions are an invasive surgery to remove the ball of the femur—or a total hip replacement—which is expensive and traumatic for the dog.
The condition occasionally affects humans, but the trigger causing the blood loss and degeneration remains a mystery despite years of research. Some have proposed that a clotting disorder or a prior injury may be to blame.
"It's a bit of a blind alley," said Todhunter, who is an orthopedic surgeon and professor of surgery at the Cornell University College of Veterinary Medicine. "By the time we see the problem, the disease has progressed to the stage where you have a clinical problem, so whatever triggered it a few months before might not be there anymore."
Previously, Todhunter and his collaborator, Dr. Jessica Hayward, a research associate in Biomedical Sciences at Cornell who specializes in canine genetics, searched canine genomes to find genes that contribute to the disease. They located a region on canine chromosome 6 that appears to be related. Their analysis included DNA from 85 small and mixed-breed dogs with Legg-Calve-Perthes disease and 102 healthy dogs provided by a colleague at Clemson University.
Now, with the additional funding from the Cornell Riney Canine Health Center, Todhunter and Hayward hope to narrow in on the exact mutation or mutations on chromosome 6 that put dogs at risk of developing the condition. They will almost double the number of dog genomes in their analysis through samples provided by a colleague at the University of Helsinki.
If the work is successful, then a genetic test can be developed to screen dogs who are carriers for the disease. This type of test could help breeders make better breeding decisions, resulting in fewer affected dogs.
The results may also provide hints to which genes in humans contribute to Legg-Calve-Perthes disease, or to a common mechanism responsible for triggering the condition in both species.
Todhunter says it is challenging to find federal funding for canine research unless the dogs serve primarily as a model for human disease. He appreciates the Cornell Riney Canine Health Center's support to continue this work, which focuses purely on improving the health and wellbeing of our canine companions.
Written by Patricia Waldron
Catching up to human treatments for canine AML
 Losing a dog to cancer is always devastating, but losing one to acute myeloid leukemia (AML) is an especially heart-wrenching experience because it happens so fast. Dr. Kelly Hume, an associate professor in the Department of Clinical Sciences and veterinary oncologist at the Cornell University Companion Animal Hospital, recalls a recent case where a three-year-old large-breed dog lived just 19 days after the initial diagnosis, despite supportive care and two different types of chemotherapy.
Losing a dog to cancer is always devastating, but losing one to acute myeloid leukemia (AML) is an especially heart-wrenching experience because it happens so fast. Dr. Kelly Hume, an associate professor in the Department of Clinical Sciences and veterinary oncologist at the Cornell University Companion Animal Hospital, recalls a recent case where a three-year-old large-breed dog lived just 19 days after the initial diagnosis, despite supportive care and two different types of chemotherapy.
Some dogs are too sick at diagnosis even to attempt treatment, but for others, the standard therapy may give them a few more months. "One of the challenges with cancer treatment is that it's such a roll of the dice," said Hume. "That's why research is so important — so that we can have better treatment guidelines and give owners more accurate expectations for the future."
Currently, when dogs receive a diagnosis of AML, they have only a fraction of treatment options available to humans. Dr. Tracy Stokol, a professor in the Department of Population Medicine and Diagnostic Sciences and veterinary clinical pathologist at the Cornell College of Veterinary Medicine, wants to give dogs and their owners more information and better choices for fighting this aggressive disease. She received a grant from the Cornell Richard P. Riney Canine Health Center to investigate what the presence of different cancerous mutations reveals about how best to approach each dog's case.
AML is a type of cancer that starts in the bone marrow and leads to the production of vast numbers of immature blood cells that ultimately crowd out the other normal cells in the blood. The cancer progresses quickly—healthy dogs suddenly become tired, stop eating, have enlarged lymph nodes or develop a fever. "It's devastating when you have this really happy dog, some of which are quite young, and then suddenly they develop an incurable disease, which is so rapidly fatal," Stokol said.
Stokol has long been fascinated by AML, starting with her training in veterinary clinical pathology, but the disease really hit home when a close family member died from AML. The death inspired her to pursue research on the canine disease more vigorously. "We're so far behind in veterinary medicine," said Stokol. "We're using drugs that are old in human medicine and it would be nice to be able to expand our treatment options."
The center funding will enable Stokol to analyze blood samples from 20 dogs diagnosed with AML along with collaborators at the University of Georgia, the University of Guelph and the University of Tennessee, veterinarians from specialty clinics, and a pathologist who specializes in human AML at the University of North Carolina at Chapel Hill. They plan to compare DNA from the cancerous cells in the blood to DNA from a cheek swab to identify new mutations. Ultimately, they hope to link the mutations to different subtypes of AML, enabling genetic testing that could indicate whether newer drugs may be more effective than the standard treatment.
With the initial work funded by the Cornell Riney Canine Health Center, Stokol also recently received a grant from the American Kennel Club Canine Health Foundation that will extend the project for another two years and include 46 additional dogs.
Stokol is eager to recruit additional dogs. Veterinarians treating dogs suspected to have AML can contact her to be considered for the study, where they will receive complimentary diagnostic testing. The study may help future AML patients to live longer lives.
"I think we can do better than what we're doing now," Stokol said, "and at least give the owners more time with their dogs."
Written by Patricia Waldron