Cytology (fluid samples only)
We offer cytologic evaluation of multiple fluid types, including body cavity fluids, cerebrospinal fluid, synovial fluid and tracheal and bronchoalveolar washes. Fluids from cystic masses and other sites (urine, bile) may also be submitted. Slide-only cytology samples (eg. FNAs from lymph nodes, solid masses and internal organs) are not accepted.
Collection and Handling
- Most fluids for cytologic exam should be submitted in an EDTA (purple top) tube. A red-top tube should be submitted if any chemistry tests (e.g. urea nitrogen, triglycerides, glucose) or culture are also requested from the fluid. Urine can be submitted in a red-top tube for cytology.
- Freshly made, air-dried, unstained smears should be submitted with the fluid. This helps minimize changes which occur with fluid storage (e.g. cell lysis, bacterial overgrowth) and can impact interpretation
- Unstained, unfixed, and unoiled smears are preferred. If submitting pre-stained smears, please do not oil or place coverslips on them (so they can be re-stained if necessary).
- Label all samples with patient identification and site/fluid sampled. Use pencil to label slides because ink may wash away during staining. Use ink to label tubes. Please do not place adhesive labels on the slides. If smears from more than one fluid are submitted, e.g. pleural and pericardial fluid, label each slide as to which site they represent.
- Avoid exposure of any cytologic specimens to formalin. Cytologic samples should be mailed in a separate container from formalin-fixed samples (formalin leaches through capped or screw-top lids and affects staining quality).
- Store the fluid samples in the refrigerator and submit with ice packs to keep cool (not in direct contact with the ice to prevent cell lysis). Do not refrigerate smears. Submit as soon as possible to the laboratory.
Tips on Preparing Smears
- Most smears can be made from un-concentrated fluid (called direct smears). Urine smears should be made from a concentrated/centrifuged sample (called sediment smears). Sediment smears can also be prepared from any fluid you suspect is poorly cellular. Please do not concentrate the entire sample (just centrifuge a portion) and indicate on any fluid tube and smears if the sample has been concentrated.
- Use a blood smear technique for most fluids (eg. body cavity effusions). The "line" technique (depicted below) can be substituted for urine or poorly cellular fluids. Use the "squash" technique (depicted below) for mucoid or viscous fluids (tracheal wash, joint fluid, bile).
- Rapidly air-dry the smears.
- Indicate on the request form that smears have been submitted with the fluid (to ensure we are aware of their presence).
Squash Technique
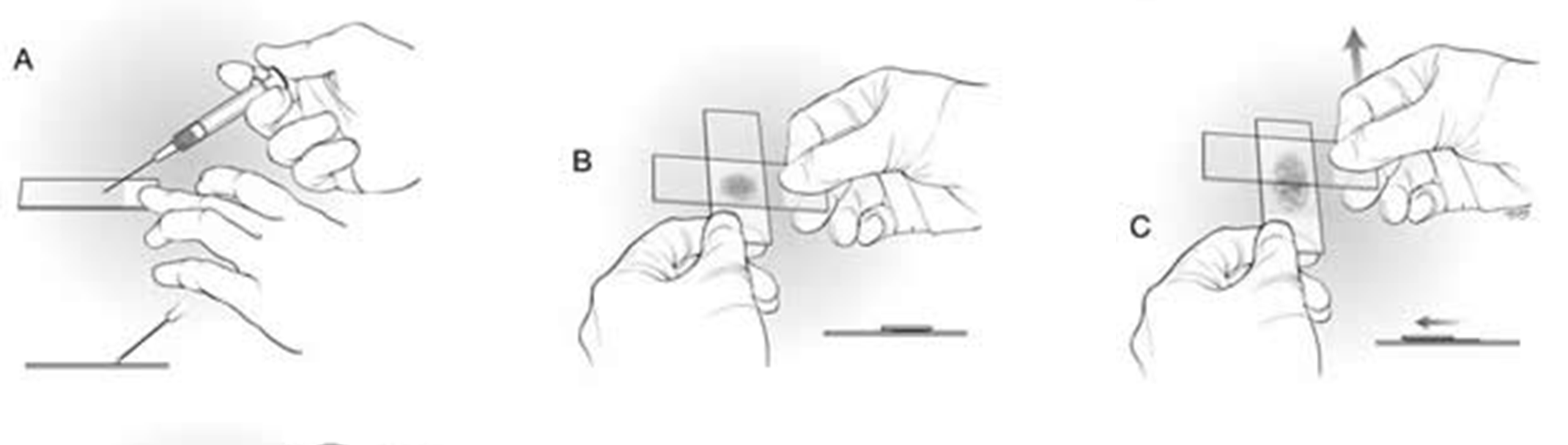
Place a drop of fluid near the frosted end of the slide. Gently place a second (spreader) slide on top of the drop and slowly pull the spreader slide across the first slide. DO NOT use excess force to spread the material, only the weight of the spreader slide is needed.
Submissions
All fluid cytology submissions must come with a completed request form - cytology submission form.
Please use one cytology submission form per patient
- One form can be used for multiple samples or tests from an individual patient
Sample Submitted/Test Requested
- For cerebrospinal taps, specify if you have collected the fluid from the atlanto-occipital (AO or cervical) or lumbosacral (LS) region.
- For tracheal washes please indicate if the sample was collected via transtracheal aspiration (TTW) or whether it was collected through an endoscope (scope).
- For urine, please indicate the method of collection (voided, catheter, cystocentesis). Urine cytology is generally used to characterize the morphology of epithelial cells, especially when neoplasia is suspected. For other urine investigations, consider a routine urinalysis.
History/Clinical Information
- Please provide a brief summary of the relevant historical, physical exam, imaging and/or bloodwork findings (ie. tell us why you are submitting this sample).
'Other diagnostic tests requested' field
- Use this field to order other Clinical Pathology or Diagnostic Laboratory tests, e.g. ordering a urinalysis with a urine cytology or a culture with a tracheal wash cytology.
Reported Results (reporting varies by fluid type)
| Visual Assessments | Volume, color, turbidity |
| Total Protein | Measured by refractometry for most fluid samples. CSF protein is measured on our automated chemistry analyzer. |
| Sample Viscosity | A subjective visual assessment of the viscosity of the sample. |
| Cell Counts | Includes both a nucleated cell and red blood cell count. In most instances this is done using an automated counter. Cell counts of poorly cellular fluids (e.g. CSF, BAL) are done manually by hemocytometer. |
| Smear Description | A complete microscopic description of the sample. |
| Smear Interpretation | We will provide you with a cytologic diagnosis where possible or a list of differential diagnoses and additional tests (if indicated) to consider based on the findings. |


